26 Chapter 26 Fluid, Electrolyte and Acid-Base Balance
By Aylin Marz
Motivation.

Ensuring that patients are sufficiently hydrated is an important part of nursing care. Intake of fluids as well as ability to pump fluids (blood) to the kidneys and away from tissue spaces are all equally important. Nurses can determine a patient is dehydrated severely if they have low blood pressure, high heart rate, cold hands and feet and low urine output. In the hospital setting dehydration can be treated. The figure shows how to do a skin turgor test for assessment of dehydration. To check skin turgor: use the thumb and index fingers to pinch an area of the skin and release it (Figure 26.1) . It should instantly return to place. Try this with your classmates!
Learning Objectives
Upon completion of the work in this chapter students should be able to:
- Critically evaluate the effects of altered blood pressure, volume, and osmolarity on maintaining fluid balance given patient medical history in a case study
- Determine the effects of abnormally high salt concentration on the health of red blood cells
- Model the effects of altered carbon dioxide and bicarbonate buffer on plasma
Background.
Fluid Balance. Your body is mostly water. Body fluids are aqueous solutions with differing concentrations of materials, called solutes. An appropriate balance of water and solute concentrations must be maintained to ensure cellular functions. If the cytosol (intracellular fluid or ICF, Figure 26.2) becomes too concentrated due to water loss, cell functions deteriorate. If the cytosol becomes too dilute due to water intake by cells, cell membranes can be damaged, and the cell can burst. Fluid balance is maintained by the exchange of water and solutes between the intracellular fluid and extracellular fluid (interstitial fluid IF and plasms, Figure 26.2)
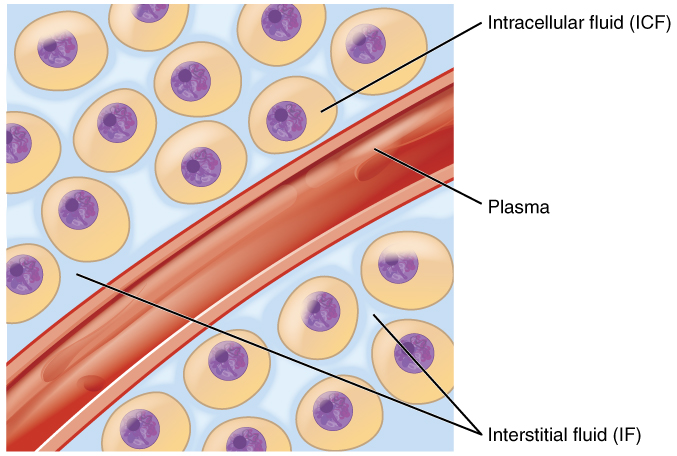
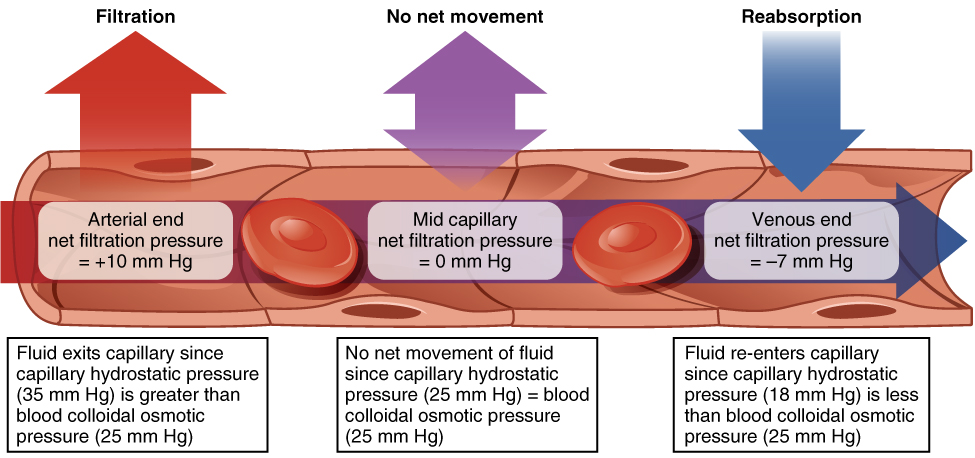
Hydrostatic pressure is the force exerted by a fluid against a wall and causes movement of fluid between compartments. In the body, blood pressure within the blood vessels correlates with hydrostatic pressure within the capillaries that are the sites of fluid exchange. Fluid can also move between compartments along an osmotic gradient (from low solute to high solute concentration) (Figure 26.3). Active transport processes require ATP to move some solutes against their concentration gradients between compartments. Passive transport of a molecule or ion depends on its ability to pass easily through the membrane, as well as the existence of a high to low concentration gradient. Passive or active processes can be used to maintain the proper solute concentrations in the extracellular and intracellular compartments.
Homeostasis requires that water intake and output be balanced. Most water intake comes through the digestive tract via liquids and food, but roughly 10 percent of water available to the body is generated at the end of aerobic respiration during cellular metabolism. Urine produced by the kidneys accounts for the largest amount of water leaving the body. The kidneys can adjust the concentration of the urine to reflect the body’s water needs, conserving water if the body is dehydrated or making urine more dilute to expel excess water when necessary. Antidiuretic Hormone ADH is a hormone that helps the body to retain water by increasing water reabsorption by the kidneys, as well as enhancing thirst to increase water intake. Other hormones that regulate electrolyte balance (see below) and neural regulation also play a role in maintaining water balance.
Electrolyte Balance. Electrolytes serve various purposes, such as helping to conduct electrical impulses along cell membranes in neurons and muscles, stabilizing enzyme structures, and releasing hormones from endocrine glands. The ions in plasma also contribute to the osmotic balance that controls the movement of water between cells and their environment. Imbalances of these ions can result in various problems in the body, and their concentrations are tightly regulated. Aldosterone and angiotensin II are two hormones that control the exchange of sodium and potassium between the renal filtrate and the renal collecting tubule. Calcium and phosphate are regulated by parathyroid hormone PTH, calcitriol, and calcitonin hormones.
Acid-Base Balance. A variety of buffering systems exist in the body that helps maintain the pH of the blood and other fluids within a narrow range—between pH 7.35 and 7.45. A buffer is a substance that prevents a radical change in fluid pH by absorbing excess hydrogen or hydroxyl ions. Most commonly, the substance that absorbs the ion is either a weak acid, which takes up a hydroxyl ion (OH–), or a weak base, which takes up a hydrogen ion (H+). Several substances serve as buffers in the body, including cell and plasma proteins, hemoglobin, phosphates, bicarbonate ions, and carbonic acid. The bicarbonate buffer is the primary buffering system of the IF surrounding the cells in tissues throughout the body. The respiratory and renal systems also play major roles in acid-base homeostasis by removing CO2 and hydrogen ions, respectively, from the body.
Pre-Laboratory Questions
- What does the term “fluid balance” refer to in the body?
- What are electrolytes?
- How does a “buffer” help maintain a particular pH?
Exercises
- Exercise 1 Fluid balance and edema: Factors affecting interstitial fluid accumulation
- Exercise 2 Electrolyte balance and red blood cells: Effects of hypernatremia and hyperchloremia
- Exercise 3 Acid-base balance and respiration: Effects of carbondioxide and buffering
Exercise 1 Fluid balance and edema: Factors affecting interstitial fluid accumulation
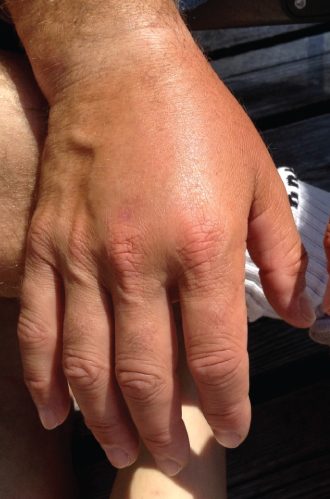
The image shows edema or swelling in the hand tissues of a patient. Edema refers to abnormal accumulation of interstitial fluid. As a nursing student you are trying to figure out what may have caused this issue.
The patient has the following history:
- Heart valve disease
- Hypertension
- Hypercalcemia (abnormally high blood calcium levels)
Question 1. For each of the conditions above, discuss how it may or may not contribute to fluid retention in the interstitial spaces. Consider fluid exchange in capillaries. The factors to consider are capillary / blood vessel health (permeability), filtration / hydrostatic pressure (blood pressure) and colloid osmotic pressure (solutes in capillary)
- Heart valve disease:
- Capillary health
- Filtration pressure
- Colloid osmotic pressure
- Hypertension:
- Capillary health
- Filtration pressure
- Colloid osmotic pressure
- Hypercalcemia:
- Capillary health
- Filtration pressure
- Colloid osmotic pressure
Question 2: In the table below indicate the anticipated effect of the deficiencies listed on factors that cause edema and edema itself. Refer to Figure 26.3 above.
| Condition | Arterial pressure (higher, lower, not changed?) | Venous pressure (higher, lower, not changed?) | Colloid osmotic pressure within capillary (higher, lower, not changed?) | Rate of water movement out of the capillary (increased, decreased, not changed?) | Probability of developing edema (increased, decreased, not changed?) |
| Leaky capillary | |||||
| Abnormally high blood volume | |||||
| Abnormally low blood pressure | |||||
| Hypocalcemia (calcium levels too low) |
Exercise 2 Electrolyte balance and red blood cells: Effects of hypernatremia and hyperchloremia
Hypernatremia refers to having blood sodium levels that are too high. Hyperchloremia refers to having blood chloride levels that are too high. In this exercise you will explore the effects of having abnormally high levels of both these ions in the environment on the health of red blood cells. Remember the process of osmosis? Consider that concept as you interpret your results.
Required Materials
- 0.9% Sodium Chloride NaCl solution
- 5% Sodium Chloride NaCl solution
- Sheep’s blood (red blood cells)
- Dropper
- Microscope slides
- Coverslips
- Microscope oil
- Compound light microscope
- Absorbent paper
- Lens paper
- Wax pen for labeling
- Gloves
Procedure
- Take two microscope slides and place them side by side on the bench. Label one of them 0.9% and the other one 5% NaCl.
- Onto each slide place one drop of the corresponding NaCl solution.
- Place one drop of Sheep’s Blood onto each slide.
- Gently place a coverslip on top of the blood solution.
- Examine each slide under the microscope. First start at low magnification to find the cells and make your way stepwise to the highest magnification of 100x objective with oil.
- Compare the shape of the red blood cells in 0.9% to those 5% NaCl solution. Describe the differences in shape.
- Which of these solutions do you think is isotonic to the red blood cell cytoplasm? Explain
- In the space below, draw the shape of the red blood cells observed at high magnification (100x) with 0.9% NaCl and with 5% NaCl.


Exercise 3 Acid-base balance and respiration: Effects of carbondioxide and buffering
Carbon dioxide CO2 is produced in aerobic respiration of cells and is present in the air we breathe inhale and exhale. The amount of this gas in the bloodstream contributes to the pH of blood since it becomes carbonic acid (CH2O3) when dissolved in water.
CO2 + H20 ↔ CH2O3
Sodium bicarbonate (same chemical that is baking soda) Na+HCO3– acts as one of the buffers in blood plasma to maintain the blood pH within the narrow limit of 7.35 and 7.45 that can support life
CO2 + H20 ↔ CH2O3 ↔ HCO3– + H+
In this exercise you will explore the effects of carbon dioxide and the buffer sodium bicarbonate on pH.
Required Materials
- Distilled water
- Plastic cups or beakers (3 per group)
- Straws
- Carbonated water
- Baking soda (sodium bicarbonate)
- Stirrer
- pH strips
Procedure
- Add distilled water into a cup and measure its pH using the pH strips. Dip the strip into the water and examine the color. Compare the color to the colors listed on the tube to determine pH. Record the pH of water:_____
- Repeat 1 for a cup full of carbonated water. Record the pH of carbonated water: ______
- Compare the pH of the distilled water to the carbonated water.
- Stir in a small amount of baking soda (sodium bicarbonate) into both the cups. Measure the pH and record it.
- pH or water+baking soda:______
- pH of carbonated water+baking soda:_______
- Now add distilled water to a third cup and use a straw to blow air into the water to push your expiratory gases into the water.
- Immediately measure the pH of the water with your expiratory gases in it. Record the pH of water plus exhaled air: _______
Post-laboratory Questions
- In Exercise 1 you determined some factors that can cause edema. The following are some treatment that are used to relieve/treat edema. For each of them explain how it works at the capillary level. Does it affect hydrostatic pressure, colloid osmotic pressure and how does that help relieve edema
-
- Lowering the patient’s blood pressure with medication:
- Increasing the patient’s protein intake:
- Prescribing a diuretic to increase the kidneys’ water output:
- In Exercise 2 you determined the effect of hypernatremia on red blood cells.
-
- Based on your results above, articulate the effect of hypernatremia on red blood cells.
- Consider osmosis. Did water move into or out of the red blood cells in the hypernatremia situation (5% NaCl)?
- If someone has hypernatremia, which of the following intravenous sodium chloride solutions could help them? Hypotonic 0.45% sodium chloride, or hypertonic 3% sodium chloride? Explain.
- In Exercise 3 you examined the effects of carbon dioxide and bicarbonate buffer on pH. Based on your results, answer the following questions.
-
- Why does carbonation change the pH? Does it make the pH more acidic or basic?
- When you blow into the water you increased the level of carbon dioxide in the water. Did this make the water more or less acidic? Explain using this formula:
CO2 + H20 ↔ CH2O3 ↔ HCO3– ↔ H+
-
- How did adding bicarbonate change the pH of the water (carbonated or distilled)? What do you expect would happen if you added bicarbonate into the water with your expiratory gases? How does this buffering help maintain blood pH?

